1. Origin of the Name
As a rising star in the field of alloy materials, the birth of high-entropy alloys (HEAs) is closely linked to the concept of entropy. But what exactly is entropy? Before unveiling the mysteries of high-entropy alloys, let's first explore the world of entropy.
The concept of entropy was introduced by German physicist Rudolf Clausius in 1865. In thermodynamics, entropy is a parameter that describes the degree of disorder in a system-the greater the disorder, the higher the entropy.
To better understand this concept, let's use a physical education (PE) class as an example:
Low-entropy state: When all students wear the same uniform with only a few exceptions, stand in neat rows, and perform warm-up exercises in a compact formation, their movement range is limited, and the entropy of the system is low.
High-entropy state: When students wear a variety of outfits, disperse after the formation is dismissed, and engage in free activities, the movement range increases, and the class becomes more disordered, resulting in a high-entropy state.
By analogy, we can think of each student as an atomic unit in an alloy. When atoms are arranged in an orderly manner, similar to students standing in formation, the alloy system is in a low-entropy state. Conversely, when atoms are randomly distributed and move freely, the system reaches a high-entropy state.
Compared to conventional alloys, this newcomer to the alloy world has a higher entropy value. Therefore, materials scientists named it high-entropy alloy. In this context, "high entropy" refers to the high degree of chemical or topological disorder at the atomic scale, meaning the atomic arrangement within the alloy is highly chaotic. Now that we've introduced the concept of high-entropy alloys, let's explore how this fascinating material was discovered.
2. The Discovery of High-Entropy Alloys
The foundation for discovering high-entropy alloys was laid as early as the late 18th century. German scientist and metallurgist Franz Karl Achard conducted an innovative study in which he prepared a series of multi-component alloys containing five to seven elements. However, this groundbreaking work was largely overlooked by metallurgists worldwide.
It wasn't until 1963 that Professor Cyril Stanley Smith (Smith, 1963) noticed and reported this research. Based on existing experimental records, it is now believed that this was one of the earliest studies related to high-entropy alloys.
Unfortunately, due to a lack of attention, progress on high-entropy alloys stalled for decades. It wasn't until the 1990s that interest in high-entropy alloys was reignited. In 1993, scientists at the University of Cambridge proposed the "confusion principle," suggesting that alloys with higher entropy are more likely to form amorphous structures. Around the same time, Taiwanese scholar Yeh Jien-Wei and his team introduced an innovative alloy design concept-designing alloys with multiple principal elements and high mixing entropy, which they named high-entropy alloys.
However, scientific progress often takes time. Despite the introduction of this concept, related research findings remained unpublished for years.
Finally, in 2004, Professor Cantor from the UK conducted an experiment in which he melted a high-mixing-entropy alloy. Surprisingly, instead of forming the anticipated amorphous structure, the alloy developed multiple brittle crystalline phases. This finding contradicted the confusion principle but confirmed Yeh's design theory. This unexpected discovery officially marked the birth of high-entropy alloys.
To explain this fascinating phenomenon, Professor Zhang Yong of the University of Science and Technology Beijing provided theoretical insights, laying the foundation for further research on HEAs. Since then, high-entropy alloys have emerged as a shining star in the field of alloy materials.
As research enthusiasm grows, the concept of high-entropy alloys has rapidly expanded into related fields such as:
High-entropy ceramics
High-entropy thin films
High-entropy steels
High-entropy superalloys
Al-Mg-based high-entropy lightweight alloys
High-entropy hard alloys
3. Unique Characteristics of High-Entropy Alloys
High-entropy alloys introduce a novel perspective in alloy design, based on the configurational entropy principle. Compared to conventional alloys, HEAs feature distinctive design concepts and unique properties. Their uniqueness can be understood from the following four aspects:
(1) Composition Characteristics
HEAs incorporate multiple principal elements to introduce chemical disorder, with no single dominant element. Unlike traditional alloys, which typically have a primary base element, HEAs consist of multiple major alloying elements. Key characteristics of HEA composition design include:
1.The alloy must contain at least five different elements.
2.The atomic percentage of each element must be between 5% and 35%.
(2) Development Characteristics
As research progresses, the concept of high-entropy alloys has evolved through three main stages:
First-generation HEAs:
Consist of five or more alloying elements.
Elements are equiatomic (equal atomic ratios).
Structure: Single-phase complex solid solution alloy.
Second-generation HEAs:
Consist of four or more alloying elements.
Non-equiatomic element ratios.
Structure: Dual-phase or multi-phase complex solid solution alloy.
Third-generation HEAs:
Expanded into high-entropy thin films and ceramics.
(3) Structural Characteristics
Despite their multiple elemental components, high-entropy alloys typically form relatively simple phase structures upon solidification. A randomly dissolved solid solution is the characteristic structure of HEAs, often exhibiting:
Face-centered cubic (FCC) structures
Body-centered cubic (BCC) structures
Hexagonal close-packed (HCP) structures
Additionally, amorphous phases may also form in some HEAs.
Conclusion
High-entropy alloys represent a revolutionary advancement in the field of alloy materials. Their unique composition, novel design approach, and promising properties make them highly valuable for next-generation structural and functional materials. With ongoing research, HEAs are expected to play a critical role in applications ranging from aerospace and automotive industries to energy, electronics, and biomedical fields.
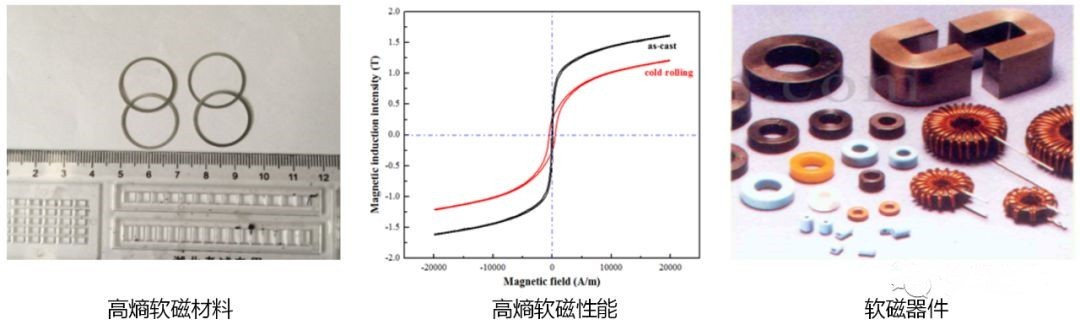
Figure 1 Phase structure of high entropy alloy
(4) Verified Performance Characteristics
Research has shown that high-entropy alloys (HEAs) possess numerous superior properties compared to traditional alloys. Currently, five major performance effects of HEAs have been scientifically confirmed:
1.Low stacking fault energy
2.Thermal stability
3.Radiation resistance
4.Corrosion resistance
5.Mitigation of the performance trade-off effect
4. Practical Applications: Where High-Entropy Alloys Shine
The unique design principles of high-entropy alloys grant them a range of outstanding properties, including excellent low-temperature mechanical performance, corrosion and wear resistance, high-temperature stability, and exceptional soft magnetic properties. These advantages give HEAs immense potential in engineering applications. Let's explore some key fields where high-entropy alloys demonstrate their remarkable capabilities.
(1) High-Entropy Soft Magnetic Materials
Soft magnetic materials are characterized by achieving maximum magnetization with minimal external magnetic fields. They are easy to magnetize and demagnetize, making them widely used in electrical and electronic equipment.
Studies have shown that certain high-entropy alloy systems exhibit excellent soft magnetic properties. Additionally, they help overcome the shortcomings of conventional soft magnetic materials, such as poor mechanical properties and unstable casting performance. As a result, HEAs hold great potential for applications in electric motors, transformers, and other industrial sectors.
(2) High-Temperature Alloy Materials
High-temperature alloys are metal materials that can operate under high temperatures and specific stress conditions for extended periods. These alloys require:
Exceptional high-temperature strength
Excellent oxidation and thermal corrosion resistance
Good fatigue resistance and fracture toughness
They are indispensable for engine hot-end components. Research indicates that high-entropy alloys exhibit remarkable thermal stability and oxidation resistance at elevated temperatures. This breakthrough offers new possibilities for developing components that operate in extreme environments, such as:
Engine turbine blades
High-temperature engineering materials
(3) Hard Tool Coatings
Hard tool coatings involve depositing a high-hardness, wear-resistant alloy thin layer onto cutting tool surfaces to protect turning, milling, planing, and grinding tools. The high hardness and strength of HEAs make them ideal candidates for such coatings, effectively enhancing tool performance and longevity.
(4) High-Entropy Photothermal Conversion Materials
Photothermal conversion refers to the process of concentrating solar radiation energy through reflection, absorption, or other mechanisms to achieve sufficiently high temperatures and meet various energy demands.
Due to the harsh service conditions of photothermal materials, they must exhibit:
Excellent high-temperature stability
Superior corrosion resistance
Low thermal expansion coefficient
Strong weather resistance
Studies have demonstrated that high-entropy alloy thin films possess outstanding corrosion resistance and high-temperature stability, making them highly promising for improving photothermal conversion efficiency and advancing solar energy applications.
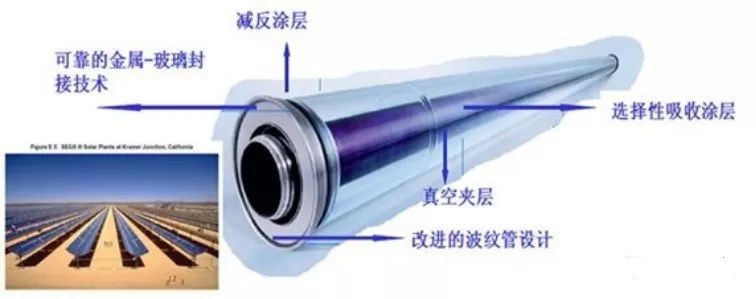
Figure 2: Heat collecting tube of photothermal converter
(5) Lightweight high entropy alloy materials
Lightweighting is an important direction for the development of future materials. In recent years, high entropy alloys have also been studied for lightweight materials and have begun to be commercialized. Common examples include mobile phone cases, mobile phone card slots and other precision parts.

Figure 3 Stamping mobile phone case
(6) Other Applications
High-entropy alloys also hold great potential in several other fields, including:
High-entropy welding materials
Low-activation high-entropy alloys
Mold materials
Catalytic materials
Semiconductor diffusion barrier layers
5. The Future of High-Entropy Alloys
How can high-entropy alloys (HEAs) be further developed? This question is a primary concern for scientists when advancing a new class of materials. Choosing the right research direction is essential for achieving maximum progress with minimal effort, enabling HEAs to play a more significant role in daily life and national defense applications.
Looking ahead, there are two key development directions that deserve the most attention:
(1) Identifying the Most Cost-Effective Performance Region
From traditional alloys to high-entropy alloys, materials have evolved along a trend of increasing entropy. However, experimental results indicate that the relationship between mixing entropy and material performance is nonlinear. In simple terms, a higher mixing entropy does not necessarily translate into better alloy performance.
Blindly pursuing higher entropy does not guarantee unlimited optimization of material properties. Moreover, as entropy increases, so does the number of constituent elements, which directly raises the production cost of the alloy. This means that focusing solely on high entropy can lead to diminishing returns, where the increase in cost outweighs the performance benefits-a scenario of "losing both the investment and the outcome."
According to statistical analyses, the most cost-effective region is not in the high-entropy alloy domain but rather at the boundary between medium-entropy alloys and high-entropy alloys. This region includes materials such as:
High-temperature alloys
Amorphous alloys
Stainless steels
Medium-entropy alloys
These materials offer a better balance between cost and performance, making this transitional region a critical area for future materials development.

Figure 4 "Cost-Effectiveness" of Alloy Materials
(2) Developing High-Efficiency Material Research Methods
High-entropy alloys (HEAs) exhibit outstanding mechanical properties, high-temperature resistance, wear resistance, and corrosion resistance, making them highly promising in various fields. However, research on high-entropy alloy thin films is still in its early stages, and their industrial application remains a work in progress. Compared to traditional alloys, HEAs have complex compositions, and their properties do not follow a linear relationship with entropy. As a result, it is impossible to design high-performance multi-component materials based solely on mixing entropy.
Material design and development is a time-consuming process, so improving research efficiency is a key factor in accelerating the advancement of HEAs.
The Role of High-Throughput Technology
In this context, the adoption of high-throughput (HT) technology becomes essential. But what exactly is high-throughput technology, and how can it accelerate material development?
If we compare alloy materials to an ocean and the development of new alloy systems to fishing, then traditional preparation methods are like fishing with a rod, where researchers can only obtain one alloy system at a time. This "one-at-a-time" approach significantly slows down material research.
Now, imagine if researchers could cast a net instead of using a fishing rod, allowing them to capture multiple alloy systems in a single attempt. This approach would greatly enhance research efficiency. High-throughput technology follows a similar principle, enabling the parallel preparation of multiple alloy systems at once, thus dramatically accelerating the development of HEAs.
A Bright Future for High-Entropy Alloys
HEAs are in a rapid growth phase, with China emerging as a major force in global research. Today, as the rising star of the materials science field, HEAs continue to attract the attention of scientists worldwide.
Although many challenges remain, they have not deterred researchers from pushing forward. As a new frontier in alloy materials, HEAs hold immense potential and promise for the future. Their mysteries are waiting to be unraveled, and their possibilities are limitless-are you ready to explore their unknown brilliance?
